|
11/27/2023 0 Comments Blueprint genetic testing 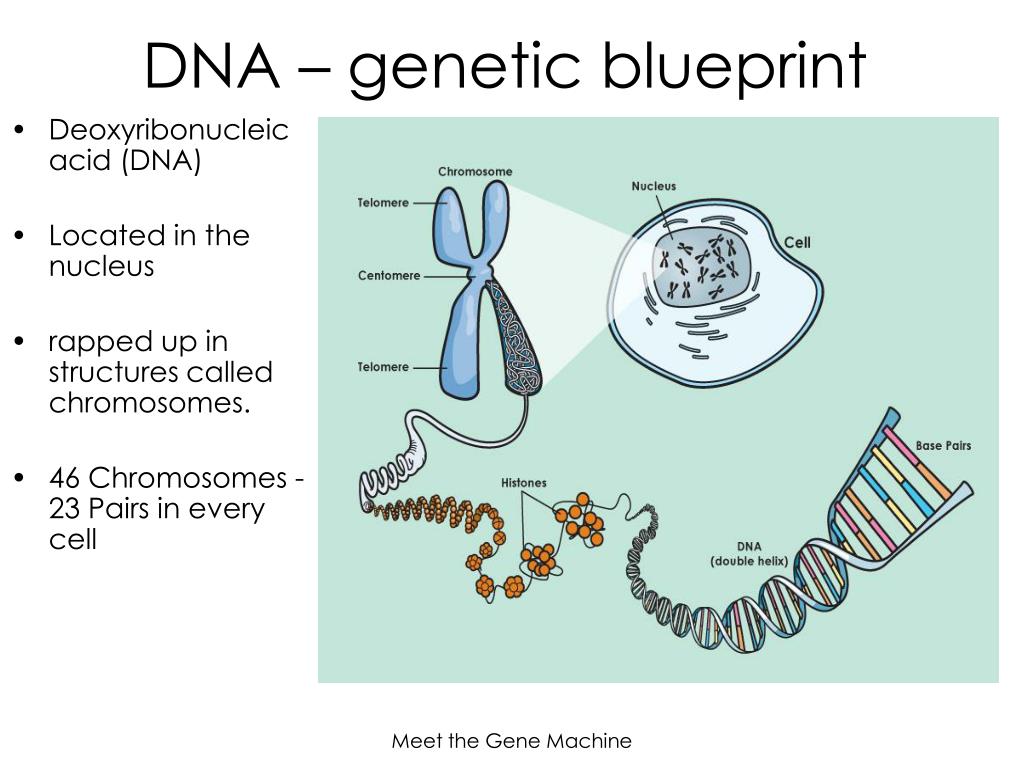
The recent entry into the marketplace of genetic tests sold directly to consumers (DTC) without external scrutiny brings added immediacy to these concerns and has renewed governmental attention to the broader question of genetic testing oversight. However, government oversight does not ensure adequately that tests can provide accurate and meaningful health information to doctors and patients, nor does it require those offering tests to disclose the evidentiary basis for the claims they make about their tests. We conclude that establishing a registry is a critical first step in the development of a more transparent, quality-centered system of oversight that will better inform and protect the public.Īs genetic testing becomes an increasingly important part of clinical practice, it is imperative that health care providers, payers, and patients in both the United States and other countries have access to high-quality tests and to information about how to order and interpret them appropriately. We discuss the sources of legal authority that empower the government to mandate that a registry be established and identify the federal agencies with the relevant expertise and resources to do so. We describe the goals of a registry, propose criteria for the inclusion of registrants and tests in the registry, and define the categories of information that should be included for such tests. We propose a ‘blueprint’ for the creation of a genetic test registry in order to expedite its implementation. A recent government recommendation for a mandatory genetic test registry has received wide stakeholder support but leaves many practical questions unanswered. Enhancing the transparency of information about what tests are being offered, for which indications tests are being offered, and the analytic and clinical validity of tests is a key prerequisite to ensuring test quality. 
Information gaps impede informed decision making by health care providers and patients. Once a client account is set up, the provider will receive a client account code and will be informed of the next steps to requesting the test and sending the specimen.While the number of genetic tests continues to grow, publicly accessible information about the analytic and clinical validity of such tests is lagging. Setting up a client account typically takes 3-5 business days.Contact for critical results and rejections.Billing contact name, email address, and phone number for invoices, associated PO# (if applicable), and address for invoices.Ordering physicians name and NPI Number.Name of the institution, address, phone number, and fax number.To request a client account, the provider must email Venessa Gamboa at the following information:.To order the test, your physician will need to create a client account with UChicago.Visit a representative physician or neurologist to see if a SCA27b genetic test is right for you.To learn more about SCA27b, check out this SCAsource article: To be or SCA27b? How to get a SCA27b test at the University of Chicago In Late May 2023, the University of Chicago Genetics Lab announced a CLIA-certified genetic test is now available. SCA27b, one of the newest identified forms of Hereditary Ataxia, is anticipated to be a highly prevalent type of Ataxia. Participants must live in the United States, be 18 years of age or older, and become a member of NAF to participate. Who is eligible?Īt this time, the Genetic Counseling and Testing Program is available to a person who is at- risk for SCA1, SCA2, and SCA3 because they have a family member with a confirmed genetic diagnosis. After counseling, if the participant and genetic counselor determine that testing is the right decision, genetic testing for SCA1, SCA2, and SCA3 will be offered at no-cost to the participant. Eligible participants will receive virtual genetic counseling at no-cost to them. NAF, in partnership with our Drug Development Collaborative, is now offering this program to overcome any obstacles that may prevent individuals from receiving genetic counseling and testing. Genetic testing is also often a requirement for participation in clinical trials for SCAs. Genetic testing can help inform decisions around family planning, healthcare/lifestyle choices, and professional/financial planning. Genetic testing to confirm a diagnosis of Ataxia can be an incredibly useful tool for persons with a family history of Ataxia. NAF is offering free virtual genetic counseling and genetic testing for individuals who are at-risk for SCA1, SCA2, or SCA3. Evaluation and Management of Ataxic Disorders (PDF).Connect Your Patient with an Ataxia Specialist.EL-PFDD Meeting for Polyglutamine Ataxias.International Congress for Ataxia Research.COVID-19 Information for Ataxia Patients.International And Other Ataxia Organizations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
